Auf dieser Seite finden Sie Antworten auf häufig gestellte Fragen im Zusammenhang mit Tests, die nicht mehr für COVID-19 verwendet und/oder verteilt werden sollten.
Dieser Abschnitt enthält Fragen und Antworten zu den Richtlinien, die in der sofort gültigen Leitlinie für klinische Laboratorien, kommerzielle Hersteller und Mitarbeiter der Food and Drug Administration beschrieben sind: Policy for Coronavirus Disease-2019 Tests during the Public Health Emergency (Revised). In diesem Abschnitt wird dieser Leitfaden als Richtlinie für Coronavirus Disease-2019-Tests bezeichnet.
Ein Verzeichnis von FAQs zu SARS-CoV-2-Tests finden Sie unter FAQs zu Tests auf SARS-CoV-2.

Q: Welche Labore, die der FDA zuvor mitgeteilt hatten, dass sie einen diagnostischen SARS-CoV-2-Test gemäß der in Abschnitt IV.A der Richtlinie für Coronavirus-Krankheit-2019-Tests dargelegten Richtlinie entwickelt und validiert haben, wurden jetzt von dieser Meldeliste gestrichen, weil die FDA entschieden hat, dass der Test nicht mehr verwendet werden sollte? (Aktualisiert am 25.9.20)
Die Labore in der folgenden Liste haben der FDA mitgeteilt, dass sie einen diagnostischen SARS-CoV-2-Test gemäß Abschnitt IV.A der Richtlinie für Coronavirus Disease-2019 Tests entwickelt und validiert haben. Obwohl die FDA sie zuvor in die Website-Meldeliste der Labore aufgenommen hatte, die diagnostische Tests im Rahmen dieser Richtlinie anbieten, wurden sie nun von dieser Meldeliste entfernt und in die nachstehende Liste aufgenommen. Wie in der Leitlinie unter anderem vermerkt, beabsichtigt die FDA, das Labor von der Benachrichtigungsliste zu streichen, wenn erhebliche Probleme mit einem solchen Test festgestellt werden, die nicht rechtzeitig behoben werden können oder wurden. Die FDA hat festgestellt, dass es signifikante Probleme mit den Tests gibt, die von den unten aufgeführten Laboren angeboten werden, die nicht rechtzeitig behoben werden können oder wurden. Daher wurden diese Labore nun von der Benachrichtigungsliste gestrichen und auf die unten stehende Streichliste gesetzt.
Die FDA erwartet, dass die Labore auf dieser Liste ihren diagnostischen, im Labor entwickelten SARS-CoV-2-Test, der zuvor auf der Benachrichtigungsliste stand, nicht mehr anbieten oder verwenden. Andere Tests, die von der EUA autorisiert sind, können von diesen Laboren in Übereinstimmung mit diesen EUAs verwendet werden.
Q: Welche kommerziellen Hersteller von Diagnosetests, die zuvor die FDA gemäß der in Abschnitt IV.C der Richtlinie für Coronavirus-Krankheit-2019-Tests beschriebenen Politik benachrichtigt hatten, wurden nun von dieser Benachrichtigungsliste gestrichen und sollten diesen Test nicht mehr anbieten? (Aktualisiert am 2.2.21)
Die in der nachstehenden Liste aufgeführten kommerziellen Hersteller haben der FDA mitgeteilt, dass sie einen diagnostischen Test gemäß Abschnitt IV.C der Richtlinie für Coronavirus-Krankheitstests 2019 validiert haben und zu vertreiben beabsichtigen. Die FDA hatte sie zuvor in die Website-Benachrichtigungsliste der kommerziellen Hersteller aufgenommen, die diagnostische Testkits im Rahmen dieser Richtlinie vertreiben, aber sie wurden nun von dieser Benachrichtigungsliste entfernt und in die unten stehende Liste aufgenommen. Wie in der Anleitung vermerkt, beabsichtigt die FDA, den Hersteller und den Test von der Benachrichtigungsliste zu streichen, wenn ein EUA-Antrag von einem kommerziellen Hersteller eines diagnostischen Tests nicht innerhalb eines angemessenen Zeitraums eingereicht wird oder wenn wesentliche Probleme mit einem solchen Test identifiziert werden, die nicht zeitnah behoben werden können oder wurden. Kommerzielle Hersteller können ihre Testnotifizierung auch freiwillig zurückziehen, und solche Tests sind mit einem Sternchen gekennzeichnet.
Die FDA erwartet, dass die Tests auf dieser Liste nicht vertrieben werden, bis eine EUA für den Test ausgestellt wird, und die FDA kann gegebenenfalls weitere Maßnahmen ergreifen.
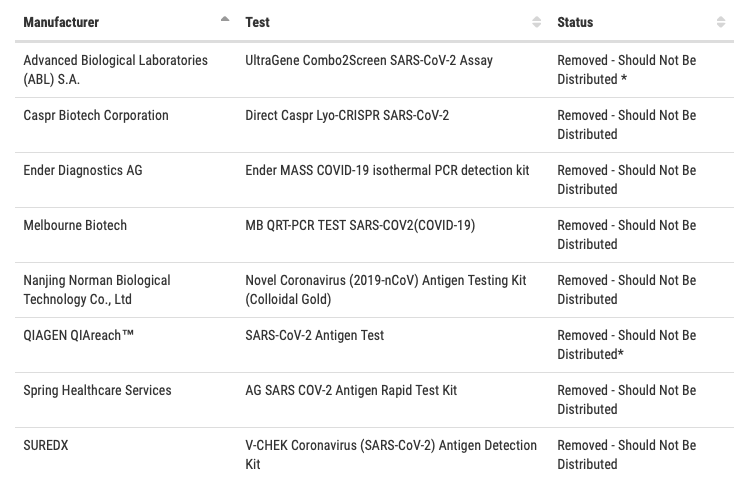
Q: Welche kommerziellen Hersteller von serologischen Tests, die zuvor die FDA gemäß der in Abschnitt IV.D der Richtlinie für Coronavirus-Krankheit-2019-Tests beschriebenen Politik benachrichtigt hatten, wurden nun von dieser Benachrichtigungsliste gestrichen und sollten diesen Test nicht mehr anbieten? (Aktualisiert 2/3/21)
Die in der nachstehenden Liste aufgeführten kommerziellen Hersteller haben der FDA mitgeteilt, dass sie einen serologischen Test gemäß Abschnitt IV.D der Richtlinie für Coronavirus-Krankheitstests 2019 validiert haben und zu vertreiben beabsichtigen. Die FDA hatte sie zuvor in die Website-Benachrichtigungsliste der kommerziellen Hersteller aufgenommen, die Serologie-Testkits im Rahmen dieser Richtlinie vertreiben, aber sie wurden nun von dieser Benachrichtigungsliste entfernt und in die unten stehende Liste aufgenommen. Wie in der Anleitung vermerkt, beabsichtigt die FDA, den Hersteller und den Test von der Benachrichtigungsliste zu streichen, wenn ein EUA-Antrag von einem kommerziellen Hersteller eines Serologie-Tests nicht innerhalb eines angemessenen Zeitraums eingereicht wird oder wenn erhebliche Probleme mit einem solchen Test festgestellt werden, die nicht zeitnah behoben werden können oder wurden. Kommerzielle Hersteller können ihre Testnotifizierung auch freiwillig zurückziehen, und solche Tests sind mit einem Sternchen gekennzeichnet.
Die FDA erwartet, dass die Tests auf dieser Liste nicht vertrieben werden, bis eine EUA für den Test ausgestellt wird, und die FDA kann gegebenenfalls weitere Maßnahmen ergreifen.
Empfehlungen für klinische Labore und Gesundheitsdienstleister in Bezug auf diese Tests finden Sie im Brief der FDA vom 19. Juni 2020 an Mitarbeiter klinischer Labore und Gesundheitsdienstleister.
| Manufacturer | Test | Status |
|---|---|---|
| Abbexa Ltd | abx294171 - COVID-19 IgG/IgM Rapid Test Kit | Removed - Should Not Be Distributed |
| Abingdon Health Ltd. | AbC-19™ Rapid Test | Removed - Should Not Be Distributed* |
| Absology Co., Ltd. | Absoludy COVID-19 IgM/IgG Combo | Removed - Should Not Be Distributed* |
| Absology Co., Ltd. | Absoludy COVID-19 IgM/IgG RAPID | Removed - Should Not Be Distributed* |
| Absology Co., Ltd. | INIST COVID-19 IgM/IgG RAPID | Removed - Should Not Be Distributed* |
| Accel Diagnostics, LLC | Rapid C2T Total Ab (IgG/IgM) Card | Removed - Should Not Be Distributed* |
| ACCOBiotech SDN.BHD | ACCO COVID-19 IgM/IgG TEST | Removed - Should Not Be Distributed |
| Aegis Sciences Corporation | SARS-CoV-2 Antibody, IgG assay | Removed - Should Not Be Distributed* |
| AESKU.DIAGNOSTICS GmbH & Co. KG | AESKULISA® SARS-CoV-2 NP IgG | Removed - Should Not Be Distributed* |
| AESKU.DIAGNOSTICS GmbH & Co. KG | AESKULISA® SARS-CoV-2 NP IgM | Removed - Should Not Be Distributed* |
| AESKU.DIAGNOSTICS GmbH & Co. KG | AESKULISA® SARS-CoV-2 S1 IgM | Removed - Should Not Be Distributed* |
| AESKU.DIAGNOSTICS GmbH & Co. KG | AESKULISA® SARS-CoV-2 S1 IgG | Removed - Should Not Be Distributed* |
| Akston Biosciences Corporation | AntiCoV-ID IgG ELISA Test | Removed - Should Not Be Distributed |
| Alfa Scientific Designs, Inc. | Clarity COVID-19 IgG/IgM Antibody Test | Removed - Should Not Be Distributed |
| Alfa Scientific Designs, Inc. | Instant-view plus COVID-19 IgG/IgM Antibody Test | Removed - Should Not Be Distributed |
| Alpha Pharma Service srl | IRIS SARS-CoV-2 IgG/IgM Rapid Test | Removed - Should Not Be Distributed |
| AngelWorld Biotech Manufacturing FZ LLC | AngelWorld V2.0 COVID-19 IgG/IgM Antibody Rapid Test | Removed - Should Not Be Distributed |
| Anhui Deepblue Medical Technology Co., Ltd. | COVID-19 (SARS-CoV-2) IgG/IgM Antibody Test Kit (Colloidal Gold) | Removed - Should Not Be Distributed |
| Ansh Labs | SARS-CoV2 IgG ELISA (AL-1001) | Removed - Should Not Be Distributed |
| Antagen Pharmaceuticals, Inc. | COVID-19 IgM/IgG Antibody Test Kit (Colloidal Gold) | Removed - Should Not Be Distributed* |
| Antibody BioPharm, Inc. | COVID-19 IgM/IgG POCT Kit (Colloidal Gold) | Removed - Should Not Be Distributed* |
| AnyGo Technology Co., Ltd | COVID-19 Capture ELISA Assay | Removed - Should Not Be Distributed* |
| Arbor Assays | DetectX SARS-CoV-2 IgG ELISA Kit | Removed - Should Not Be Distributed* |
| Arbor Vita Corporation | CoVisa IgG Test | Removed - Should Not Be Distributed |
| Artron BioResearch Inc./ Artron Laboratories Inc. | COVID-19 IgM/IgG Antibody Test | Removed - Should Not Be Distributed* |
| Asan Pharmaceutical Co., Ltd. | Asan Easy Test COVID-19 IgG/IgM | Removed - Should Not Be Distributed* |
| Atlas Link (Beijing)Technology Co., Ltd | NovaTest: One Step COVID-19 IgG/IgM rapid test | Removed - Should Not Be Distributed |
| Audacia Bioscience | CMC-19D SARS-CoV2 (COVID-19) Rapid Antibody Test | Removed - Should Not Be Distributed |
| Aurora Biomed Inc. | COVID-19 IgG/IgM Rapid Test Cassette (Colloidal Gold) | Removed - Should Not Be Distributed |
| Autobio Diagnostics Co. Ltd. | Anti-SARS-CoV-2 Rapid Test | Removed - Should Not Be Distributed |
| Axium BioResearch, Inc. | AxiumHealth COVID-19 IgG/IgM Antibody Test | Removed - Should Not Be Distributed* |
| Beijing Decombio Biotechnology Co., Ltd. | Novel Coronavirus IgM/IgG Combo Rapid Test-Cassette (Serum/Plasma/Whole blood) | Removed - Should Not Be Distributed* |
| Beijing DiaGreat Biotechnologies Co. Ltd. | 2019-nCoV IgM Antibody Determination Kit | Removed - Should Not Be Distributed* |
| Beijing DiaGreat Biotechnologies Co. Ltd. | 2019-nCoV IgG/IgM Antibody Rapid Test Kit | Removed - Should Not Be Distributed* |
| Beijing DiaGreat Biotechnologies Co. Ltd. | 2019-nCoV IgG Antibody Determination Kit | Removed - Should Not Be Distributed* |
| Beijing Kewei Clinical Diagnostic Reagent Inc. | Genonto RapidTest10 COVID-19 IgG/IgM Antibody Rapid Test Kit | Removed - Should Not Be Distributed |
| Beroni Group | SARS-CoV-2 IgG/IgM Antibody Detection Kit | Removed - Should Not Be Distributed |
| BestNovo (Jiangsu) Medical Technology Co., Ltd. | BestNovo COVID-19 IgM/IgG Antibody Rapid Test Kit | Removed - Should Not Be Distributed |
| Bethyl Laboratories, Inc. | Bethyl SARS-CoV-2 IgG IVD ELISA | Removed - Should Not Be Distributed* |
| Biobase Biodustry (Shandong) Co., Ltd. | SARS-CoV-2 IgM/IgG Antibody Test Kit (Colloidal Gold) | Removed - Should Not Be Distributed |
| Biolidics Limited | 2019-nCoV IgG/IgM Detection Kit (Colloidal Gold) | Removed - Should Not Be Distributed* |
| BioMedomics, Inc. | COVID-19 IgM-IgG rapid test | Removed - Should Not Be Distributed* |
| Biomerica, Inc. | COVID-19 IgG/IgM Rapid Test | Removed - Should Not Be Distributed |
| Bioscience Diagnostic Technology Company Ltd. | Diagnostic Kit for Novel Coronavirus (2019-nCoV) IgG Antibody | Removed - Should Not Be Distributed |
| Bioscience Diagnostic Technology Company Ltd. | Diagnostic Kit for Novel Coronavirus (2019-nCoV) IgM Antibody | Removed - Should Not Be Distributed |
| Bioscience(Chongqing) Diagnostic Technology Co., Ltd. | Qualitative Diagnostic Kit for Novel Coronavirus (2019-nCoV) IgM Antibody | Removed - Should Not Be Distributed |
| Bioscience(Chongqing) Diagnostic Technology Co., Ltd. | Qualitative Diagnostic Kit for Novel Coronavirus (2019-nCoV) IgG Antibody | Removed - Should Not Be Distributed |
| Bioscience(Tianjin) Diagnostic Technology Co., Ltd. | Qualitative Diagnostic Kit for Novel Coronavirus(2019-nCoV) IgM Antibody | Removed - Should Not Be Distributed |
| Bioscience(Tianjin) Diagnostic Technology Co., Ltd. | Qualitative Diagnostic Kit for Novel Coronavirus(2019-nCoV) IgG Antibody | Removed - Should Not Be Distributed |
| Bioscience(Tianjin) Diagnostic Technology Co., Ltd. | Novel Coronavirus (2019-nCoV) Antibody Rapid Test | Removed - Should Not Be Distributed |
| BioSure (UK) Limited | BioSURE COVID-19 Antibody Rapid Test (REF: 92001COV) | Removed - Should Not Be Distributed* |
| BioSys Laboratories, Inc. | BioSys Plus COVID-19 IgM/IgG Rapid Test | Removed - Should Not Be Distributed |
| Bloom Diagnostics AG | Bloom COVID-19 Test | Removed - Should Not Be Distributed* |
| Boston Bio Lab Inc. | Boston Bio EZ Covid-19 | Removed - Should Not Be Distributed* |
| Boston Biopharma Inc. | BB Rapid-MT | Removed - Should Not Be Distributed* |
| BreviTest Technologies, LLC | Brevitest SARS-CoV-2 IgG Test | Removed - Should Not Be Distributed* |
| BTNX, Inc. | Rapid Response™ COVID-19 IgG/IgM Test Cassette | Removed - Should Not Be Distributed* |
| Calbiotech, Inc. | ErbaLisa® COVID-19 IgG | Removed - Should Not Be Distributed |
| Camtech Diagnostics Pte Ltd | Camtech Covid-19 IgM/IgG 2C Rapid Test | Removed - Should Not Be Distributed |
| Camtech Diagnostics Pte Ltd | Camtech Covid-19 IgM/IgG 1C Rapid Test | Removed - Should Not Be Distributed |
| Cell ID Pte Ltd | Smart COVID-19 IgM/IgG Rapid Diagnostic Test (RDT) | Removed - Should Not Be Distributed* |
| Changchun Wancheng Bio-Electron Co., Ltd. | COVID-19 IgG/IgM ANTIBODY RAPID TEST KIT (Colloidal gold immunochromatography) | Removed - Should Not Be Distributed |
| Chembio Diagnostic Systems, Inc. | DPP COVID-19 IgM/IgG System | Removed - Should Not Be Distributed |
| Chemtron Biotech, Inc. | Chemtrue®Rapid COVID-19 IgM/IgG Antibody Test | Removed - Should Not Be Distributed |
| ClearSign Diagnostics Co | Novel Coronavirus (SARS-CoV-2) Antibody (IgM / IgG) Test | Removed - Should Not Be Distributed |
| ClearSign Diagnostics Co., LLC | ClearSign Diagnostics SARS-CoV-2 Antibody (IgM and IgG) Test | Removed - Should Not Be Distributed |
| Core Technology Co., Ltd. | RapidTest COVID-19 IgM/IgG Ab Test | Removed - Should Not Be Distributed |
| CoreMedica Laboratories, Inc. | Capillary Blood Collection Kit & Laboratory Analysis for SARS-CoV-2 ELISA IgG and IgM | Removed - Should Not Be Distributed |
| CTK Biotech, Inc. | OnSite® COVID-19 IgG/IgM Rapid Test | Removed - Should Not Be Distributed |
| Diabetomics, Inc | CovAbScreen™ SARS-CoV-2 Ab Test | Removed - Should Not Be Distributed* |
| DIALAB(ZJG) Biotech Co., Ltd. | SARS-CoV-2 IgG/IgM Antibody Test (Fluorescence Immunoassay) |
Removed - Should Not Be Distributed* |
| Diazyme Laboratories, Inc. | Diazyme SARS-CoV-2 Antibody Rapid Test | Removed - Should Not Be Distributed* |
| Discount Diagnostics | Global WholeHealth Partners RDT IgG/IgM Antibody test | Removed - Should Not Be Distributed |
| DNALink, Inc. | AccuFind COVID19 IgG | Removed - Should Not Be Distributed* |
| Dynamiker Biotechnology (Tianjin) Co., Ltd. | RapidCOV™ 2019-nCOV IgG/IgM Rapid Test | Removed - Should Not Be Distributed |
| EACHY Biopharmaceuticals Co., Ltd. | AccuRapid™ SARS-CoV-2 IgM/IgG Test Kit (Fluorescence Lateral Flow Immunoassay) | Removed - Should Not Be Distributed |
| EACHY Biopharmaceuticals Co., Ltd. | AccuRapid™ SARS-CoV-2 IgM/IgG Test Kit (Lateral Flow Immunoassay) | Removed - Should Not Be Distributed |
| EACHY Biopharmaceuticals Co., Ltd. | SmartScreen COVID-19 IgM/IgG Test Kit | Removed - Should Not Be Distributed |
| Edinburgh Genetics Limited | ActivXpress+ COVID-19 IgG/IgM Immunoassay Complete Testing Kit | Removed - Should Not Be Distributed* |
| EpiGentek | SeroFlash SARS-CoV-2 IgM/IgG Antibody Detection Kit | Removed - Should Not Be Distributed* |
| Epitope Diagnostics, Inc. | EDI COVID-19 Nucleocapsid IgG Quantitative ELISA Kit | Removed - Should Not Be Distributed |
| Epitope Diagnostics, Inc. | KT-1033 EDI™ Novel Coronavirus COVID-19 IgG ELISA Kit | Removed - Should Not Be Distributed* |
| Epitope Diagnostics, Inc. | KT-1032 EDI™ Novel Coronavirus COVID-19 IgG ELISA Kit | Removed - Should Not Be Distributed* |
| ET Healthcare Inc. | Pylon COVID-19 IgM/IgG Assay | Removed - Should Not Be Distributed* |
| EUROIMMUN AG | Anti-SARS-CoV-2 ELISA (IgA) | Removed - Should Not Be Distributed* |
| Fosun Pharma USA Inc. | Fosun COVID-19 IgG/IgM Rapid Antibody Detection Kit | Removed - Should Not Be Distributed* |
| GenBody Inc. | GenBody COVID-19 IgM/IgG | Removed - Should Not Be Distributed |
| Genelogic Research Limited | Genelogic COVID-19 IgM/IgG Antibody Rapid Test | Removed - Should Not Be Distributed |
| GeneScan Diagnostics, LLC | AmeriDx® SARS-CoV-2 IgG/IgM Rapid Test Kit | Removed - Should Not Be Distributed |
| GeneScan Diagnostics, LLC | AmeriDx® SARS-CoV-2 IgG/IgM-T Antibody Rapid Test Kit | Removed - Should Not Be Distributed |
| Genesprint Group, Ltd. | Genesprint Diagnostic Kit for IgG/IgM Antibody to SARS-CoV-2 test | Removed - Should Not Be Distributed* |
| Genlantis Diagnostics, Inc. | CovidQuik Coronavirus (COVID-19) IgM/IgG Antibody Test | Removed - Should Not Be Distributed* |
| Genobio Pharmaceutical Co., Ltd. | COVID-19 IgM/IgG Lateral Flow Assay | Removed - Should Not Be Distributed* |
| Getein Biotech Inc. | One Step Test for Novel Coronavirus (2019-nCoV) IgM/IgG antibody (Colloidal Gold) | Removed - Should Not Be Distributed |
| Global Go (Distributed Core Technology Co., Ltd.) | RapidTest COVID-19 IgM/IgG Ab Test | Removed - Should Not Be Distributed |
| Gold Standard Diagnostics | SARS-CoV-2 IgM ELISA Test Kit | Removed - Should Not Be Distributed |
| Gold Standard Diagnostics | INgezim® COVID 19 CROM Rapid Test Kit | Removed - Should Not Be Distributed* |
| Gold Standard Diagnostics | INgezim® COVID 19 DR ELISA Test Kit | Removed - Should Not Be Distributed* |
| Goldsite Diagnostics Inc | SARS-CoV-2 IgG/IgM kit | Removed - Should Not Be Distributed* |
| GREEN ENERGY TECHNOLOGY SOLUTIONS, LLC | AMP Rapid Test SARS-CoV-2 IgG/IgM | Removed - Should Not Be Distributed* |
| Guangdong Hecin Scientific, Inc. | SARS-CoV-2 IgM Antibody Rapid Test Kit | Removed - Should Not Be Distributed* |
| Guangzhou Decheng biotechnology | DOCHEK™ SARS-CoV-2 Antibody Rapid Test Kit | Removed - Should Not Be Distributed* |
| Guangzhou Fenghua Bioengineering Co., Ltd. | SARS-CoV-2 IgG/IgM Rapid Test | Removed - Should Not Be Distributed |
| H&Z Life Science Co. Ltd. | Anti-COVID-19 Virus IgM/IgG Test Kit (lateral flow assay) | Removed - Should Not Be Distributed |
| H-Guard (China) Co., Ltd. | Novel Coronavirus COVID-19 IgM/IgG Test Kit (colloidal gold) | Removed - Should Not Be Distributed* |
| Hangzhou AllTest Biotech Co., Ltd. | AllTest 2019-nCoV IgG/IgM Rapid Test Cassette | Removed - Should Not Be Distributed |
| Hangzhou AllTest Biotech Co., Ltd. | AllTest COVID-19 IgG/IgM Rapid Test Strip | Removed - Should Not Be Distributed |
| Hangzhou Clongene Biotech Co., Ltd. | COMBRA COVID-19 IgM/IgG Rapid Test Cassette | Removed - Should Not Be Distributed |
| Hangzhou Realy Tech Co Ltd. | 2019-nCOV IgG/IgM Rapid Test | Removed - Should Not Be Distributed |
| Hangzhou Sejoy Electronics & Instrument Co., Ltd. | COVID-19 IgG/IgM Rapid Test Cassette | Removed - Should Not Be Distributed* |
| Hangzhou Testsea Biotechnology Co., Ltd. | One Step SARS-CoV2(COVID-19) IgG/IgM Test | Removed - Should Not Be Distributed* |
| Horizon Pharmaceuticals, Inc. | Horizon RDT SARS CoV-2 IgM/IgG Antibody Test | Removed - Should Not Be Distributed |
| Hunan RunKun Pharmaceutical Co., Ltd. | SARS-CoV-2 lgM/lgG Test Kit (Colloidal Gold) | Removed - Should Not Be Distributed* |
| ICT international | 2019-nCoV IgG/IgM Rapid Test Cassette | Removed - Should Not Be Distributed* |
| IMMY, Inc. | clarus SARS-CoV-2 Total Antibody EIA | Removed - Should Not Be Distributed* |
| Innovation Biotech (Beijing) Co., Ltd. | SARS-COV-2 IgM/IgG Antibody Rapid Test (Immunochromatographic Method) | Removed - Should Not Be Distributed |
| Invenio Medical | COVID-19 IgG/IgM Rapid Test Cassette (WB/S/P) | Removed - Should Not Be Distributed |
| Invenio Medical | COVID-19 IgG/IgM Ab Rapid Test | Removed - Should Not Be Distributed |
| Iontox | COVID-19 (SARS-CoV-2) IgG antibody test kit | Removed - Should Not Be Distributed* |
| Jiangsu Dablood Pharmaceutical Co, Ltd. | AssuranceAB™ COVID-19 IgM/IgG Rapid Antibody Test | Removed - Should Not Be Distributed* |
| Jiangsu Dablood Pharmaceutical Co. Ltd. | COVID-19 IgM/IgG Rapid Test | Removed - Should Not Be Distributed |
| Jiangsu Eubo Biotechnology Co., Ltd. | EUBO COVID-19 IgG/IgM Rapid Test Cassette (WB/S/P) | Removed - Should Not Be Distributed |
| Jiangsu Macro & Micro-Test Med-Tech Co., Ltd. | SARS-CoV-2 IgM/IgG Rapid Assay Kit (Colloidal Gold) | Removed - Should Not Be Distributed* |
| Jiangsu Superbio Biomedical (Nanjing) Co., Ltd. | SARS-CoV-2 (COVID-19) IgM/IgG Antibody Fast Detection Kit (Colloidal Gold) | Removed - Should Not Be Distributed* |
| Jiangsu Superbio Biomedical (Nanjing) Co., Ltd. | ThermoGenesis SARS-CoV-2(COVID-19) IgM/IgG Antibody Fast Detection Kit (Colloidal Gold) | Removed - Should Not Be Distributed* |
| JOYSBIO (Tianjin) Biotechnology Co., Ltd. | COVID-19 IgG/IgM Rapid Test Kit (Colloidal Gold) | Removed - Should Not Be Distributed |
| Lepu Medical Technology (Beijing) Co., Ltd. | Lepu SARS-CoV-2 Antibody Rapid Test | Removed - Should Not Be Distributed |
| Leritas LLC | DualTec SARS-CoV-2 Test Kit | Removed - Should Not Be Distributed* |
| Lifeassay Diagnostics (Pty) Ltd | Test-it COVID-19 IgM/IgG Lateral Flow Assay | Removed - Should Not Be Distributed |
| LumiQuick Diagnostics, Inc. | QuickProfile™ 2019-nCoV IgG/IgM Antibody Test | Removed - Should Not Be Distributed |
| Maccura Biotechnology Co., Ltd. | Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) IgM/IgG Antibody Assay Kit by Colloidal Gold Method | Removed - Should Not Be Distributed* |
| MedicalSystem Biotechnology Co., Ltd. | Coronavirus Disease 2019 Antibody (IgM/IgG) Combined Test Kit | Removed - Should Not Be Distributed* |
| MedMira Inc. | REVEALCOVID-19™ Total Antibody Test | Removed - Should Not Be Distributed* |
| Melbourne Biotech | MB COVID-19 Antibody Detection Kit | Removed - Should Not Be Distributed |
| Mokobio Biotechnology R&D Center | SARS-CoV-2 IgM & IgG Quantum Dot Immunoassay | Removed - Should Not Be Distributed* |
| Mokobio Biotechnology R&D Center Inc | COVID-19 IgM & IgG Rapid Test (Colloidal Gold Assay) | Removed - Should Not Be Distributed* |
| Multi-G bvba | COVID-19 IgG/IgM Rapid Test Cassette | Removed - Should Not Be Distributed* |
| Nanjing Norman Biological Technology Co., Ltd | Novel Coronavirus (2019-nCoV) IgG/IgM Antibody Testing Kit (Colloidal Gold) | Removed - Should Not Be Distributed* |
| Nanjing SYNTHGENE Medical Technology Co., Ltd. | SYNTHGENE COVID-19 IgM/IgG Rapid Detection Kit (Colloidal Gold Method) | Removed - Should Not Be Distributed |
| Nanjing Vazyme Medical Technology Co., LTD. | 2019-nCoV IgG / IgM Detection Kit (Colloidal Gold-Based) | Removed - Should Not Be Distributed* |
| Nanjing Weiyun Biotechnology Co., Ltd. | SARS-CoV-2 (COVID-19) IgM/IgG Antibody Fast Detection Kit (Colloidal Gold) | Removed - Should Not Be Distributed |
| Nano-Ditech Corporation | Nano-Check COVID-19 IgG/IgM Antibody Test | Removed - Should Not Be Distributed* |
| NanoEntek, Inc. | FREND COVID-19 IgG/IgM Duo | Removed - Should Not Be Distributed* |
| NanoResearch, Inc. | NanoMedicina™ SARS-COV-2 IgM/IgG Antibody Rapid Test | Removed - Should Not Be Distributed |
| Nantong Diagnos Biotechnology Co., Ltd. | (2019-nCoV) New coronavirus Antibody Test (Colloidal Gold) | Removed - Should Not Be Distributed |
| Nantong Egens Biotechnology Co., Ltd | EGENS COVID-19 IgG/IgM Rapid Test Kit | Removed - Should Not Be Distributed |
| Naturitious LLC | Viralert COVID-19 IgG/IgM ANTIBODY RAPID TEST KIT | Removed - Should Not Be Distributed* |
| Newscen Coast Bio-Pharmaceutical Co., Ltd. | COVID-19 Microfluidic Combo Test | Removed - Should Not Be Distributed |
| Newscen Coast Bio-Pharmaceutical Co., Ltd. | COVID-19 IgG/IgM Rapid Test | Removed - Should Not Be Distributed |
| Oncosem Onkolojik Sistemler San.ve Tic. A.S | CRT (COVID-19 Rapid Test) | Removed - Should Not Be Distributed* |
| One Milo, Inc. | InstantRapid® SARS CoV-2 IgG IgM Test | Removed - Should Not Be Distributed |
| Otogenetics | OTO-Artron COVID-19 IgM/IgG Antibody Rapid Detection test | Removed - Should Not Be Distributed* |
| Pacific Connect Group LLC (US) | SARS-CoV-2 IgG/IgM Rapid Test Kit | Removed - Should Not Be Distributed |
| PCL Inc. | COVID19 IgG/IgM Rapid Gold | Removed - Should Not Be Distributed |
| Pellecome LLC (Distributed Core Technology Co., Ltd.) | CoreTest COVID-19 IgM/IgG Ab Test | Removed - Should Not Be Distributed |
| PerGrande BioTech Development Co., Ltd. | SARS-CoV-2 Antibody Detection Kit (Colloidal Gold Immunochromatographic assay) | Removed - Should Not Be Distributed* |
| Phamatech | COVID19 IgG / IgM Rapid Test | Removed - Should Not Be Distributed* |
| Pinnacle Biolabs | SARS-CoV-2 IgG/IgM Rapid Test | Removed - Should Not Be Distributed |
| Plexense, Inc. | ACCEL ELISA COVID-19 | Removed - Should Not Be Distributed* |
| Profarma, UAB | ProRapid Coronavirus Disease (SARS-CoV-2) IgG/IgM Rapid Test Cassette Single Use Kit | Removed - Should Not Be Distributed |
| Promedical | COVID-19 Rapid Test | Removed - Should Not Be Distributed |
| Promedical Equipment Pty Ltd | Promedical IgM/IgG Rapid Detection Kit | Removed - Should Not Be Distributed |
| Prometheus Bio Inc. | 2019-nCoV IgG/IgM Test (Whole blood/Serum/Plasma) Colloidal Gold | Removed - Should Not Be Distributed* |
| ProteomeTech Inc. | KOVIcheck COVID-19 IgG/IgM | Removed - Should Not Be Distributed* |
| Pure Genetic Medical Ltd. (Distributed Boson Biotech Ltd. Co) | Rapid 2019-nCoV IgG/IgM Combo Test Card | Removed - Should Not Be Distributed |
| RayBiotech Life, Inc. | Novel Coronavirus (SARS-CoV-2) IgM and IgG Antibody Detection Kits for Serum | Removed - Should Not Be Distributed |
| RayBiotech Life, Inc. | Novel Coronavirus (SARS-CoV-2) IgM and IgG Dual Combined Antibody Detection Kit for Serum | Removed - Should Not Be Distributed |
| Reszon Diagnostics International Sdn. Bhd. | RESZON (COVID-19) (Sars-Cov-2) IgG/IgM Antibody Test | Removed - Should Not Be Distributed* |
| Reszon Diagnostics International Sdn. Bhd. | Reszon COVID-19 Rapid IgG/IgM Test | Removed - Should Not Be Distributed* |
| Ring Biotechnology Co Ltd | COVID-19 IgM/IgG Rapid Test Kit | Removed - Should Not Be Distributed* |
| Safecare Biotech (Hagzhou) Co., Ltd. | SAFECARE COVID-19 IgG/IgM Rapid Test Device | Removed - Should Not Be Distributed |
| Saladax Biomedical | COVID-19 IgG/IgM Rapid Antibody Test | Removed - Should Not Be Distributed* |
| ScheBo® • Biotech AG ScheBo® | SARS-CoV-2 Quick | Removed - Should Not Be Distributed* |
| Sciteck Diagnostics | COVID19 Automated Antibody Assay | Removed - Should Not Be Distributed |
| SD Biosensor | STANDARD Q COVID-19 IgM/IgG Duo | Removed - Should Not Be Distributed |
| Sensing Self Pte. Ltd. | COVID-19 Rapid IgM/IgG Combined Antibody Assay Pre-Screening Kit | Removed - Should Not Be Distributed |
| Shandong ThinkLab Biotechnology Co., Ltd. | 2019-nCoV IgM/IgG antibody Test Kit (Colloidal-gold Assay) | Removed - Should Not Be Distributed |
| Shanghai Eugene Biotech Co., Ltd. | SARS-CoV2 (COVID-19) IgG/IgM Rapid Test | Removed - Should Not Be Distributed |
| Shanghai Fosun Long March Medical Science Co., Ltd. | Fosun COVID-19 IgG/IgM Rapid Antibody Detection Kit | Removed - Should Not Be Distributed* |
| Shanghai Outdo Biotech Co., Ltd. | Novel Coronavirus (SARS-CoV-2) Antibody (IgM / IgG) Test | Removed - Should Not Be Distributed* |
| Shenzen Landwind Medical Co., Ltd. | COVID-19 IgG/IgM Rapid Test | Removed - Should Not Be Distributed* |
| Shenzhen Lvshiyuan Biotechnology Co., Ltd | COVID-19 (2019-nCoV) Coronavirus IgG/IgM Rapid Test Kit | Removed - Should Not Be Distributed |
| Shijiazhuang Hipro Biotechnology Co., Ltd. | COVID-19 IgM/IgG Antibody Test Kit (Colloidal Gold) | Removed - Should Not Be Distributed |
| Singuway Biotech Inc. | COVID-19 IgG/IgM Detection Kit (Colloidal Gold) | Removed - Should Not Be Distributed |
| Sinocare, Inc. | SARS-CoV-2 Antibody Test Kit (Colloidal Gold Method) | Removed - Should Not Be Distributed |
| Sober Holdings, LLC | ProMed Rapid Testing - COVID-19 IgG/IgM Test Kit | Removed - Should Not Be Distributed |
| Sober Holdings, LLC | ProMed Rapid Test | Removed - Should Not Be Distributed |
| Spartacus-Biomed | LFA COVID-19 IgG & IgM Rapid Test Device | Removed - Should Not Be Distributed |
| Spring Health Care AG | COVID-19 IgG/IgM Rapid Test Cassette (Whole Blood/Serum/Plasma) | Removed - Should Not Be Distributed |
| Sugentech, Inc. | SGTi-flex COVID-19 IgM/IgG | Removed - Should Not Be Distributed* |
| Sunbeam Laboratories, LLC. | Sunbeam Laboratories Covid-19 Rapid Test Kit | Removed - Should Not Be Distributed* |
| Sure Bio-tech | API Covid-Rapid IgM/IgG Antibody Test Kit | Removed - Should Not Be Distributed |
| Sure Bio-Tech | API Pharma Covid-Rapid IgM/IgG Antibody Test | Removed - Should Not Be Distributed |
| Suzhou Kangheshun Medical Technology Co., Ltd | SARS-CoV-2 IgG/IgM Rapid Test Cassette | Removed - Should Not Be Distributed |
| Telepoint Medical Services | SARS-CoV-2 IgG/IgM Rapid Qualitative Test | Removed - Should Not Be Distributed |
| TheraTest Laboratories, Inc. | EL-Anti-SARS-CoV-2 IgG | Removed - Should Not Be Distributed* |
| Tianjin Beroni Biotechnology Co. Ltd | SARS-CoV-2 IgG/IgM Antibody Detection Kit | Removed - Should Not Be Distributed |
| Tianjin New Bay Bioresearch Co., Ltd. | Quikpac II COVID-19 IgG/IgM | Removed - Should Not Be Distributed |
| TM Testing, Inc. | TM Test Kits, CoVID-19 Rapid Antibody Serology Test | Removed - Should Not Be Distributed |
| Top Biotech Sdn. Bhd | TOP RAPID COVID-19 Rapid Antibody IgG/IgM Test Kit | Removed - Should Not Be Distributed |
| Truvian Sciences Inc. | Easy Check COVID-19 IgM/IgG™ POC Antibody Test | Removed - Should Not Be Distributed* |
| Universal Meditech Inc. | DiagnosUS® SARS-CoV-2 Antibody (IgG/IgM) Test | Removed - Should Not Be Distributed |
| Velox Biosystems | COVID-19 Microarray IgG Immunoassay | Removed - Should Not Be Distributed* |
| VicTorch Meditek Inc. | COVID-19 Serology IgM ELISA | Removed - Should Not Be Distributed* |
| VicTorch Meditek Inc. | COVID-19 Serology IgG ELISA | Removed - Should Not Be Distributed* |
| VicTorch Meditek Inc. | COVID-19 IgG/IgM Rapid test Cassette | Removed - Should Not Be Distributed* |
| Victory Square Health Canada, Inc. | Safetest-COVID19 IgG / IgM - Elisa Kit | Removed - Should Not Be Distributed* |
| Victory Square Health, Inc. | Safetest Covid-19 IgG/IgM Rapid Test | Removed - Should Not Be Distributed* |
| Virotech Diagnostics GmbH | Gold Standard Diagnostics SARS-CoV-2 IgA ELISA Test Kit | Removed - Should Not Be Distributed* |
| VITA Testing | COVID-19 IgM/IgG Antibody Rapid Test Kit | Removed - Should Not Be Distributed |
| Vitro Diagnostics | Vitro Diagnostics COVID 19 IgG/IgM Rapid Test Cassette Serology Device | Removed - Should Not Be Distributed |
| VivaChek Biotech (Hangzhou) Co., Ltd. | VivaDiag COVID-19 IgM/IgG Rapid Test | Removed - Should Not Be Distributed |
| Vivera Pharmaceuticals, Inc. | Vivera Pharma COVID-19 Rapid Test | Removed - Should Not Be Distributed* |
| Vivera Pharmaceuticals, Inc. | COVx-RT | Removed - Should Not Be Distributed |
| W.H.P.M., Inc. | COVID-19 IgM/IgG Rapid Test | Removed - Should Not Be Distributed |
| W.H.P.M., Inc. | COVISURE™ COVID-19 IgM/IgG Rapid Test | Removed - Should Not Be Distributed |
| Wuhan EasyDiagnosis | Ediagnosis COVID-19 (SARS-CoV-2) IgM/IgG Antibody Test | Removed - Should Not Be Distributed |
| Wuhan UNscience Biotechnology Co., Ltd. | Coronavirus-19 (COVID-19) Antibody (IgM/IgG) Rapid Test Kit (Colloidal gold immunochromatography) | Removed - Should Not Be Distributed |
| Wuhu 3H Biotechnology Co. Ltd. | COVID-19 IgG/IgM Test Kit (Colloidal Gold Method) | Removed - Should Not Be Distributed |
| Xiamen AmonMed Biotechnology Co., Ltd. | Helix-19 COVID-19 IgM/IgG Test Kit (Colloidal Gold) | Removed - Should Not Be Distributed |
| Xiamen AmonMed Biotechnology Co., Ltd. | COVID-19 IgM/IgG test kit (Colloidal Gold) | Removed - Should Not Be Distributed* |
| Xiamen Wiz Biotech Co., Ltd. | Diagnostic Kit (Colloidal Gold) for IgG/IgM Antibody to SARS-CoV-2 | Removed - Should Not Be Distributed |
| Zalgen Labs, LLC. | ReSARS CoV-2 (N) IgG ELISA | Removed - Should Not Be Distributed* |
| ZEUS Scientific, Inc. | ZEUS Rapid SARS-CoV-2 IgM/IgG Test System | Removed - Should Not Be Distributed* |
| Zhejiang Orient Gene Biotech, Co., Ltd. | COVID-19 IgG/IgM Rapid Test Cassette | Removed - Should Not Be Distributed |
| Zhengzhou Fortune Bioscience Co., Ltd. | COVID-19 Antibody Rapid Test Kit (Colloidal Gold Immunochromatography Method) | Removed - Should Not Be Distributed |
| Zhengzhou Fortune Bioscience Co., Ltd. | COVID-19 IgM Antibody Rapid Test Kit | Removed - Should Not Be Distributed |
| Zhengzhou Fortune Bioscience Co., Ltd. | COVID-19 IgG Antibody Rapid Test Kit | Removed - Should Not Be Distributed |
| Zhongshan Bio-Tech Co Ltd. | SARS-CoV-2 IgM/IgG (GICA) | Removed - Should Not Be Distributed* |
| Zhuhai Encode Medical Engineering Co., Ltd | Novel Coronavirus (COVID-19) IgG/IgM Rapid Test Device | Removed - Should Not Be Distributed* |
| Zhuhai Keyu Biological Engineering Co., Ltd. | SARS-CoV-2 IgG/IgM Rapid Test Kit | Removed - Should Not Be Distributed |
| Zhuhai Livzon Diagnostics, Inc. | Diagnostic Kit for IgM/IgG Antibody to Coronavirus (SARS-CoV-2) (Colloidal Gold) | Removed - Should Not Be Distributed* |
Übersetzt mit www.DeepL.com/Translator aus fda.gov